Thematic studies
News
Case studies
“Patient-centric”: new strategies to pursue for companies in the healthcare and packaging industry

With the rise of the Internet, patients have better access to information and can therefore compare different treatments and medicines. Moreover, social networks are allowing patients and caregivers to communicate and share their experiences. With more information, more investment and growing communities: patients have become more “smart” in their consumption and are changing the game in the pharmaceutical industry. The effectiveness of the product is no longer the only challenge. Patients now expect low cost, more personalised, better services with better results. Moreover, the advent of the Internet of Things and the growing need for the management of data has given rise to newcomers to the healthcare market: Health IT. The major historic players in pharmacy and biotechnologies only have a short amount of time to rethink their approach and response to today’s patient expectations.
The pharmaceutical group AstraZeneca has organised workshops with key players to define what the “patient-centric” approach represents. This proposal has been validated by patients from the online community of PatientsLikeMe:
“ Putting the patient at the heart of an open-ended commitment, respecting human suffering and giving, with empathy, the best experience and the best outcome for this person and their family ”.
This definition places the emphasis on five key criteria:
- Including the patient in the related approaches,
- Sharing the objectives centred around the patient and their family,
- Allowing patients to take control of their health,
- Working in a respectful and open way,
- Working hand in hand, as partners.
The aim is to provide a better therapeutic approach as a result of the understanding of the patient with an experience and optimal results for patients and their family.
The patient: from passive recipient to active partner
- Emergence of the patient role
For a long time the patient was a passive recipient of medical care without being involved in research. It is only since the adoption of the Nuremberg Code in 1947 that the patient had to give their voluntary and informed consent when they are the subject of research.
Increasing numbers of patients are now active partners in defining and prioritising research. Consequently, the development of drugs has a clear, organised and flexible process, which takes patient feedback much more into account than ever before. Moreover, the FDA now makes sure that the participation of patients is an integral part of the decision-making process: drug development patient consultation programmes (DDPCP), patient representative programmes (PRP), etc. The patient has moved from the status of “subject" to "consumer".
- Digitalisation and awareness
Patients and caregivers are more engaged with healthcare providers. Thanks to social networks, they are sharing their experiences with other patients who are looking to exchange information about their case or find out more about the different treatments available.
59%* of companies have made “patient centricity” an absolute priority in 2018. In this regard, they are implementing strategies of co-creation, studying every patient pathway in depth, better organising care and aiming for smart use of data and connected devices. This is a real challenge for manufacturers of pharmaceutical products.
Breaking the traditional pharmaceutical model
- Developing a patient-centric culture
Being “patient-centric” is not as easy as it seems. Firstly, it is about changing the existing order and, consequently, making some adjustments within teams which will help employees understand the holistic experience of patients.
Pharmaceutical companies must therefore instil a new culture which incorporates the fact that success is only possible as a result of cooperation. In this regard, most companies have already started to implement a patient-centric programme. It is more essential than ever to bridge the gap separating biopharmaceutical professionals from people living with a disease.
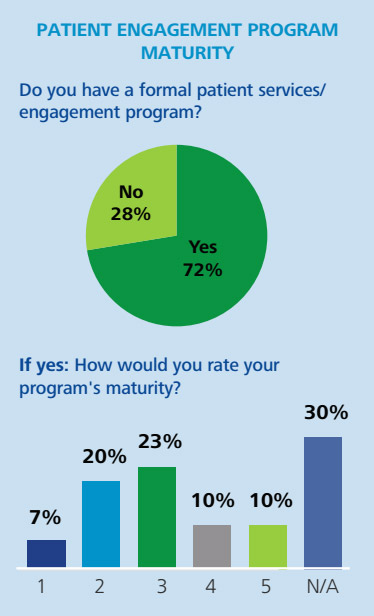
- Adopting a more personalised approach
With the global digital transformation of industries and patients that are very active on the internet, moving to an ecosystem centred around the patient represents the promotion of a new state of mind. Companies in the healthcare sector are used to communicating with patients in an impersonal way, notably through mass media campaigns.
- Implementing new strategies
Certain newcomers to the sector (wearables, mobile applications, Health IT, etc.), which are located outside of the traditional market, are rapidly evolving to meet the new expectations of patients. Pharmaceutical companies must rapidly reflect and draw up new strategies to improve their “patient-centric” approach to:
- Automate processes and optimise the use of digital technology,
- Implement collaborative relationships with patients, paymasters, healthcare professionals and regulatory bodies,
- Establish a key partnership allowing the pharmaceutical industry to access advanced or highly personalised platforms that bring them closer to patients and their expectations,
- Provide educational support (medical and pharmacovigilance information, nurse training support, care between visits, etc.),
- Create patient-centric groups to better support patients in every step of their care pathway (patient meetings, advisory committees, etc.).
“Patient-centric” approach and insulated packaging
Being patient-centric also means providing solutions which create a reliable supply chain: this guarantees that patients get the right medicines, at the right time and under the right conditions.
The “patient centric” approach has a significant impact on the pharmaceutical cold chain and the management of marketing the products. It is about:
- Reinforcing patient acceptance of treatment and ensuring satisfaction
- Simplifying the transportation of treatments by patients
- Guaranteeing that the product will be maintained at the correct temperature range
- Allowing patients to go on holiday when they want, with complete peace of mind
Insulated packaging solutions for medicinal products are designed in collaboration with patients, experts in temperature-controlled packaging and pharmaceutical marketing services. This design begins with discussions so as to understand the treatment and the needs of the patient. Insulated packaging must not only guarantee the security and efficacy of the product (environmental contamination, product stability, shocks, light, etc.), but also integrate requirements such as:
- Being easy to open and close
- Providing instructions and advice
- Facilitating quality control
- Being adapted to caregivers and/or patients with limited manual dexterity, gripping force or diminished eyesight.
The global design of an insulated packaging solution must therefore also include the physiological, physical and social characteristics of the targeted patients. So, patient-centric innovation in insulated packaging solutions now exceeds the requirements of the FDA in order to offer a better user experience.
" We live in a world increasingly shaped by human intervention where design can enable or disable people."Clarkson and Coleman, 2015